SRN-901 undergoes a more rigorous test than the typical company announcement. Rather than stopping at a headline lifespan figure, the paper combines survival data with frailty scoring and multiple layers of molecular analysis. The result is a broader argument that SRN-901 not only helps mice live longer, but also shifts several aging-related systems in a healthier direction.
That distinction matters because longevity research faces a recurring problem: many compounds appear promising when judged by a single endpoint, but lose force when researchers ask harder mechanistic questions. Aging is not a single malfunction, but a cascade involving nutrient sensing, inflammation, mitochondrial decline, cellular cleanup, and metabolic drift. A drug intended to broadly affect aging therefore has to show more than one kind of benefit, and this study is structured to make exactly that case.
A Multi-Pathway Approach to Aging
SRN-901 is a combination therapy built from multiple molecules. These components were selected to influence pathways tied to mTOR inhibition, autophagy, mitophagy, senescent cell clearance, inflammation, energy production (NAD+), and stress resistance. That design reflects a central idea in modern aging biology: single-target interventions often struggle against a process as networked and complex as aging.
Several of these pathway terms can sound abstract, so they are worth translating before turning to the results. mTOR is a nutrient-sensing growth pathway often linked to aging. Autophagy is the cell’s internal recycling system. Mitophagy is the selective removal of damaged mitochondria. NAD+ is a molecule central to energy metabolism and cellular repair. In plain language, SRN-901 is designed to quiet overactive growth and inflammatory signals while improving cellular cleanup, mitochondrial quality control, and metabolic resilience.
A Late-Life Lifespan Test
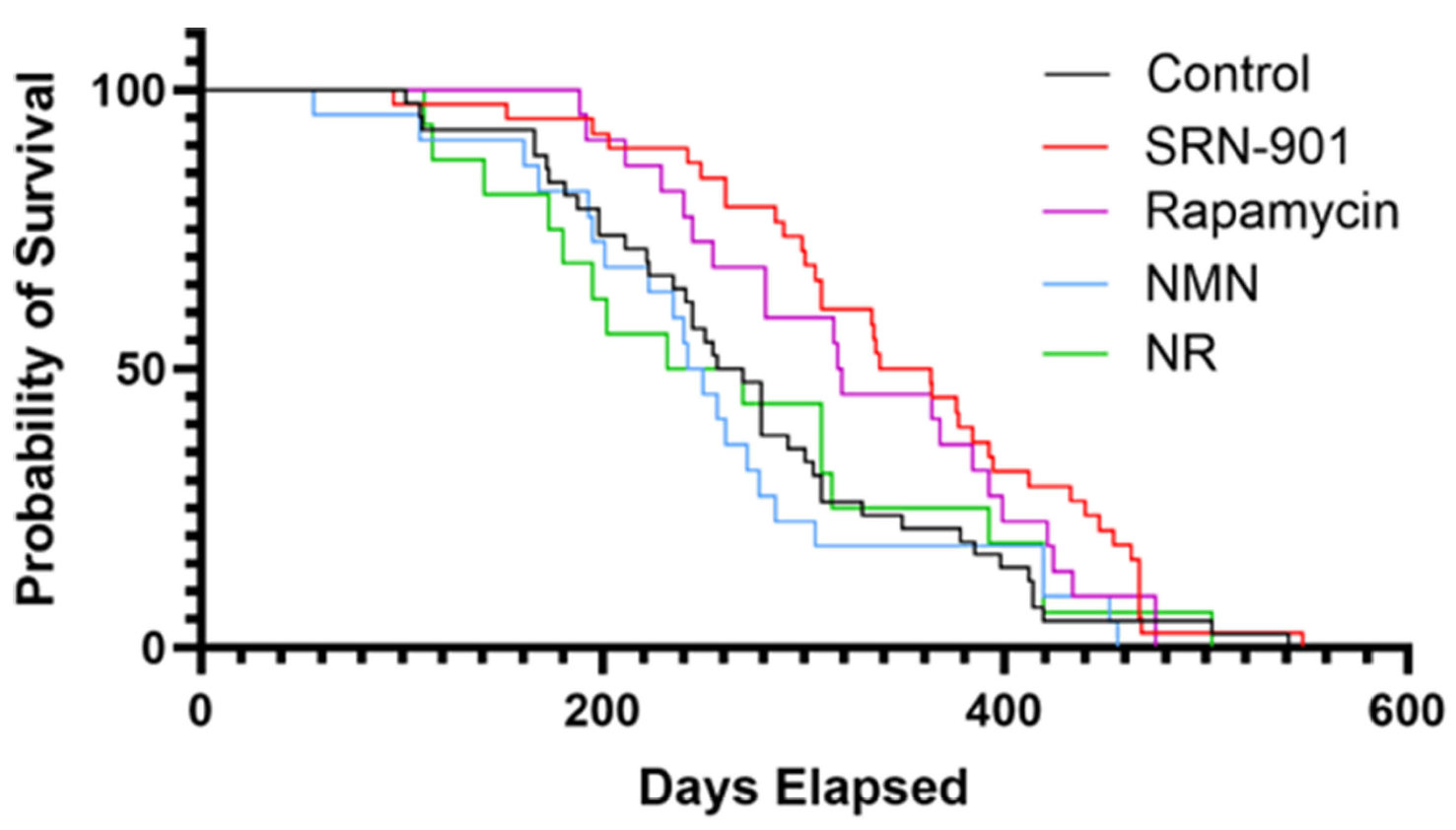
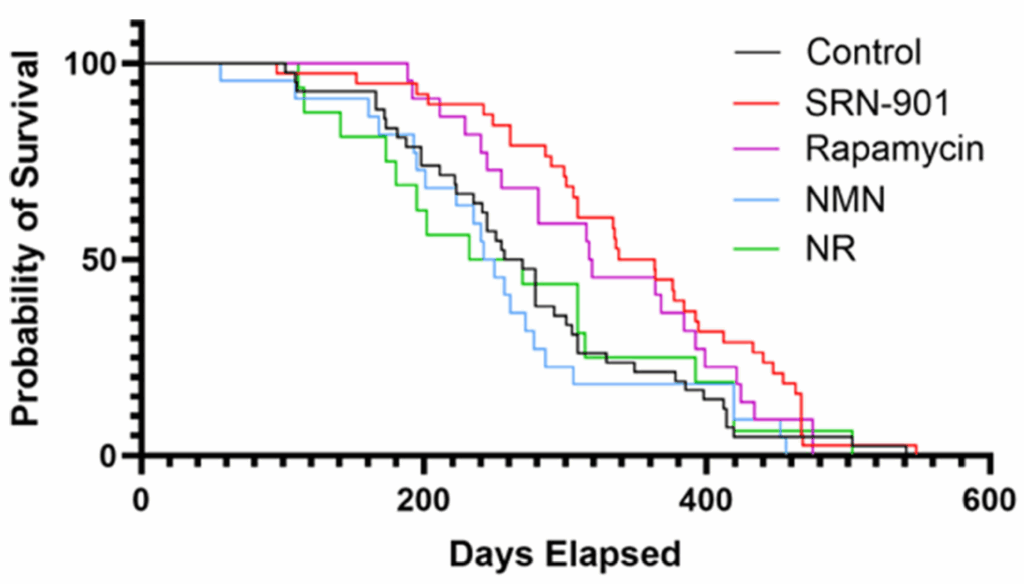
The study was designed as a late-life intervention rather than a youth-preserving experiment. Researchers treated 18-month-old C57BL/6 mice, an age the paper describes as roughly analogous to late-middle-aged to older adult humans, and compared SRN-901 with placebo as well as benchmark agents rapamycin, nicotinamide mononucleotide (NMN), and nicotinamide riboside (NR). That setup matters because many anti-aging interventions weaken when treatment begins after age-related damage is already underway.
SRN-901 increased median remaining lifespan from 263.5 days in placebo-treated mice to 350.5 days, a 33% gain, and reduced the hazard of death by 46%, with a hazard ratio of 0.54. Rapamycin also improved survival, though less strongly, while NMN and NR did not significantly outperform placebo in this experiment. That is the kind of result that moves a longevity claim from merely interesting to more difficult to dismiss, especially because the intervention began late in life rather than early.
Healthy Lifespan Extension
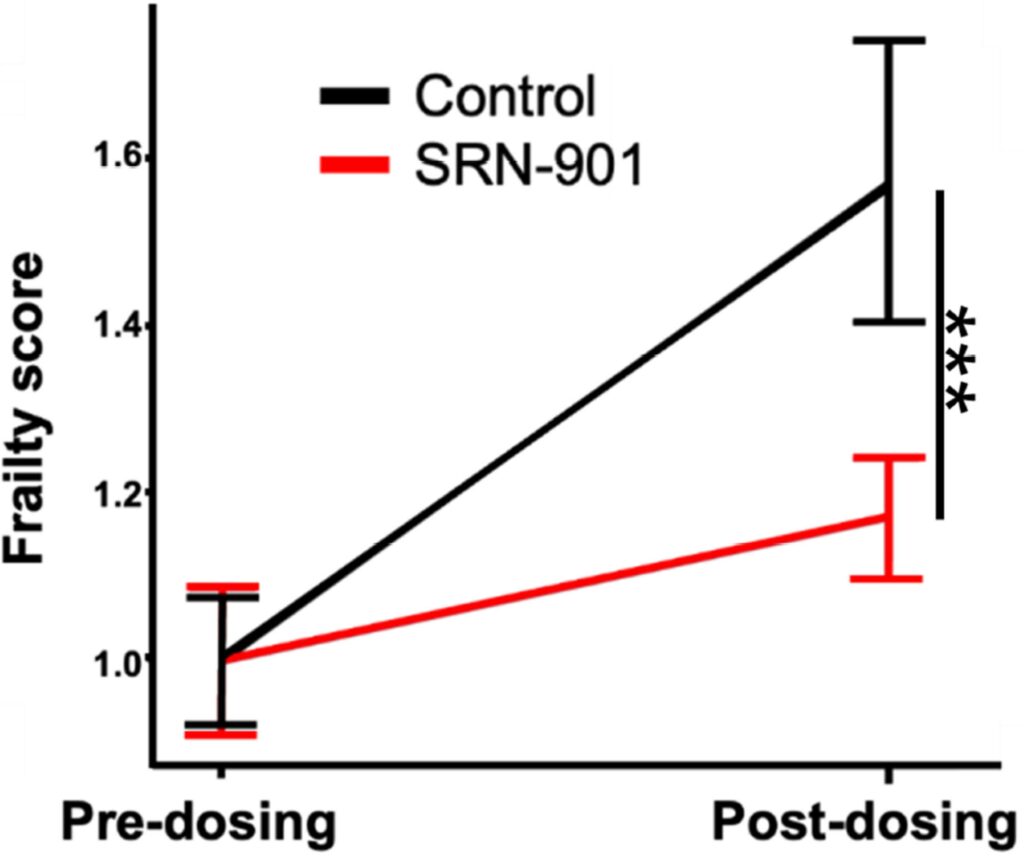
Living longer is not the same as aging well, so the paper also tracks frailty, a composite measure of visible and functional decline across multiple body systems. Investigators used a validated mouse frailty index that scores signs such as posture, grooming, musculoskeletal decline, and other non-invasive health deficits. This is important because it asks whether the added time was biologically useful rather than simply prolonged.
After 128 days of treatment, baseline-normalized frailty rose to 1.57 in the placebo group but only to 1.17 in the SRN-901 group, which the authors calculate as a 70% attenuation of frailty progression. Post-mortem examinations also found lower tumor incidence in treated mice, 13.2% versus 19.0% in controls, though that finding should be read as supportive rather than as proof that SRN-901 is an anti-cancer therapy. Taken together, these data strengthen the paper’s central argument, suggesting that the survival benefit tracked with broader physiological preservation.
What the Gene Expression Data Suggest
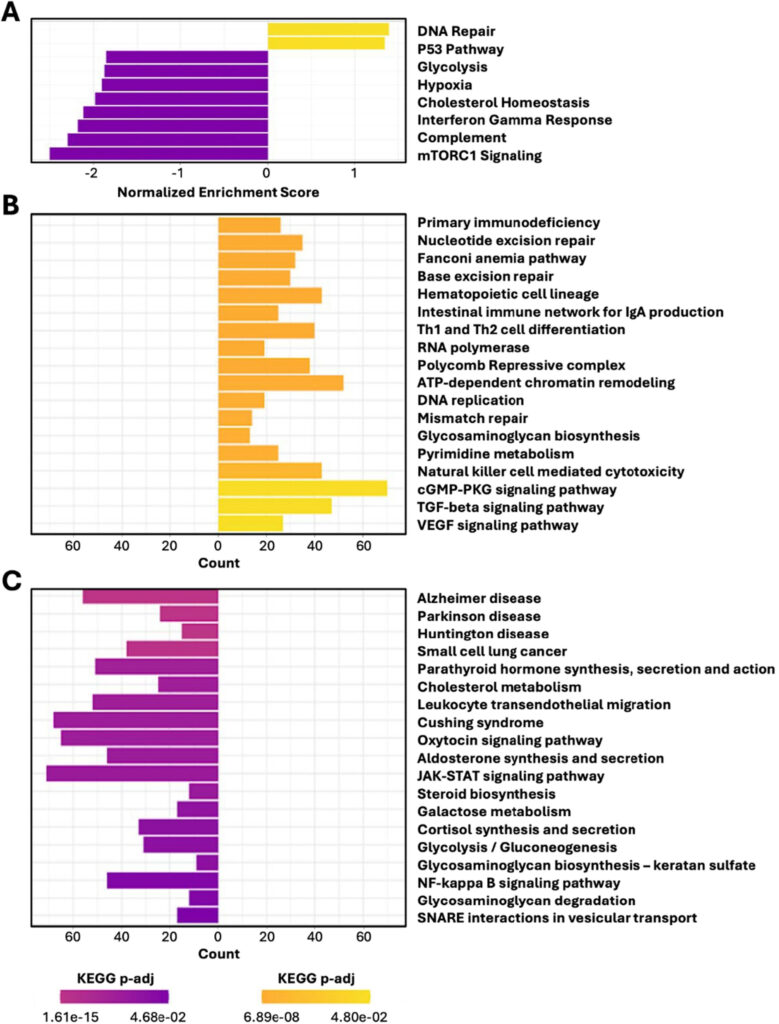
To determine whether those outward benefits were matched by deeper biological change, the researchers performed RNA sequencing on whole-blood samples. RNA sequencing measures which genes are more or less active, while gene-set enrichment analysis looks for coordinated shifts across entire pathways rather than isolated genes. Put differently, it moves the story from phenotype, what the mice looked like, to mechanism, what their cells appeared to be doing differently.
Compared with placebo, SRN-901 was associated with higher activity in DNA repair, autophagy, and mitophagy pathways, and lower activity in pathways tied to apoptosis, PI3K/AKT/mTOR signaling, interleukin-6/Janus kinase/signal transducer and activator of transcription 3 inflammatory signaling, nuclear factor kappa B inflammation, insulin resistance, and reactive oxygen species. The paper also found downregulation of an Alzheimer disease pathway annotation, but explicitly cautions that this should be interpreted as pathway-level overlap in whole blood, not as evidence of cognitive improvement or disease modification. That caveat strengthens the manuscript’s credibility by showing that the authors distinguish molecular resemblance from clinical proof.
Metabolism and the Chemistry of Aging
The metabolomics portion of the study asks a different question: how did the chemistry of the blood change with treatment? Metabolomics tracks small molecules produced or consumed by metabolism, and those molecules can reveal whether an organism is drifting toward oxidative stress, impaired glucose handling, or other age-associated imbalances. This is the layer that helps explain how a longevity intervention might reshape day-to-day physiological function.
The authors report that age-related metabolic shifts seen in control mice were blunted or reversed in treated mice, with 78 metabolites described as rescued by SRN-901 and the overall profile moving closer to that of younger animals. Pathway analysis highlighted glutathione metabolism, a major antioxidant defense network, alongside changes in metabolites related to liver and inflammatory biology. This matters because oxidative stress is one of the recurring mechanisms through which researchers believe cells accumulate damage over time.
Proteomics Adds Another Layer
A final layer came from proteomics, the large-scale measurement of proteins circulating in the blood. If transcriptomics shows what cells appear poised to do, proteomics helps show what the biological system is actually producing in functional molecular machinery. That makes it a valuable bridge between gene expression and organism-level outcomes such as frailty and survival.
The proteomic analysis profiled more than 10,000 protein groups, carried 5,023 proteins into statistical analysis, and identified 47 proteins with significant changes between placebo and SRN-901 groups. Pathway enrichment then pointed to coordinated shifts in tryptophan metabolism, lysine degradation, arachidonic acid metabolism, succinate signaling during inflammation, butyrate metabolism, starch and sucrose metabolism, and branched-chain amino acid degradation, while phenylalanine and tyrosine metabolism moved downward. In practical terms, that pattern argues against a one-off artifact and toward broader remodeling of inflammatory, energetic, and nutrient-handling systems.
What This Study Does and Does Not Prove
The paper does not prove human efficacy. It does not isolate which ingredient contributes most to the effect, and it does not erase the translational gap that has humbled many aging interventions before clinical testing. What it does do, clearly and credibly, is elevate SRN-901 from an intriguing longevity claim to a manuscript-backed, multi-pathway candidate that now warrants closer investigation.
With peer-reviewed validation now in place, SRN-901 moves beyond early-stage claims. Taken together, the lifespan, frailty, transcriptomic, metabolomic, and proteomic data make the manuscript more compelling than the average preclinical longevity paper. The study does not rely on a single spectacular graph, but on a convergence of signals that point in roughly the same direction: less physiological decline, lower mortality risk, and molecular changes consistent with healthier aging biology. That is the strongest version of the case for SRN-901, and why this manuscript carries more weight than a simple promotional update.