Main Points
- A classical cell-death protein, MLKL, drives stem cell aging without killing cells
- Aging hematopoietic stem cells accumulate mitochondrial damage via MLKL activation
- The mechanism is metabolic and structural, not primarily genetic or transcriptional
- This reframes how aging interventions might target dysfunction, not just cell removal
For years, biologists treated certain proteins as irreversible switches. Once flipped, the outcome was assumed: the cell dies.
MLKL was one of those switches. It sat at the end of a well-mapped pathway, executing necroptosis, a form of inflammatory cell death. Its role seemed final, even definitive.
But a 2026 study complicates that certainty. It shows that MLKL can activate, move inside the cell, and quietly degrade its machinery without killing it. The result is not absence, but persistence in a diminished state.
Why This Matters
Aging is often described as loss, but many systems fail not because cells disappear, but because they remain and function poorly. Hematopoietic stem cells are a prime example.
These cells sustain blood and immune production across a lifetime, yet with age they become biased, less regenerative, and metabolically constrained. That shift contributes directly to immunosenescence and chronic inflammation.
This study suggests that part of that decline is actively driven by a stress-response pathway that damages cells without eliminating them. This reframes aging as, in part, a problem of persistent dysfunction rather than simple attrition.
The Problem No One Could Fully Explain
Hematopoietic stem cell aging has long been recognized but poorly unified mechanistically. Researchers have identified multiple contributing factors, including inflammation, replication stress, mitochondrial decline, and DNA damage.
Each of these processes correlates with aging, yet none fully explains how stem cells transition from functional to impaired states. The field has lacked a clear pathway linking upstream stress signals to downstream dysfunction.
Previous work implicated the RIPK3–MLKL axis, but interpretation remained ambiguous. Was this pathway killing stem cells, or doing something more subtle? This paper directly resolves that uncertainty by separating activation from outcome.
A Protein with the Wrong Job Description
MLKL is canonically defined as an executioner protein. Once activated by RIPK3, it disrupts membranes and induces necroptosis. That framework leaves little room for intermediate states.
However, biological systems often repurpose pathways depending on context. The researchers asked whether MLKL activation in aging stem cells might diverge from its classical function.
Their findings suggest exactly that. MLKL activates under stress, but instead of completing the death program, it localizes to mitochondria and alters cellular physiology. This represents a functional shift rather than a binary on-off outcome.
What the Researchers Actually Did
The study used mouse hematopoietic stem cells across young and aged cohorts, applying stressors that mimic aging-related insults. These included inflammatory stimulation, replication stress, and repeated transplantation challenges.
Genetic models were central to the design. The researchers used MLKL knockout mice, RIPK3 knockout mice, and phosphorylation-deficient MLKL variants to isolate pathway-specific effects.
They combined this with a multi-layered methodological approach. Live biosensors tracked MLKL activation, electron microscopy assessed mitochondrial structure, and metabolic assays measured ATP production and glycolytic flux. This integration allowed them to connect signaling, structure, and function.
What They Found
The first key result is that MLKL activation increases with age and stress in hematopoietic stem cells. This activation is dependent on RIPK3 and becomes more frequent in aged populations.
Critically, this activation does not lead to increased cell death. Stem cell numbers remain stable, and markers of apoptosis or necroptosis do not significantly change. This indicates that MLKL is operating outside its canonical role.
Instead, MLKL drives functional decline. Stem cells exhibit increased myeloid bias, reduced lymphoid output, and diminished regenerative capacity. When MLKL is absent, these aging-associated phenotypes are partially reversed.
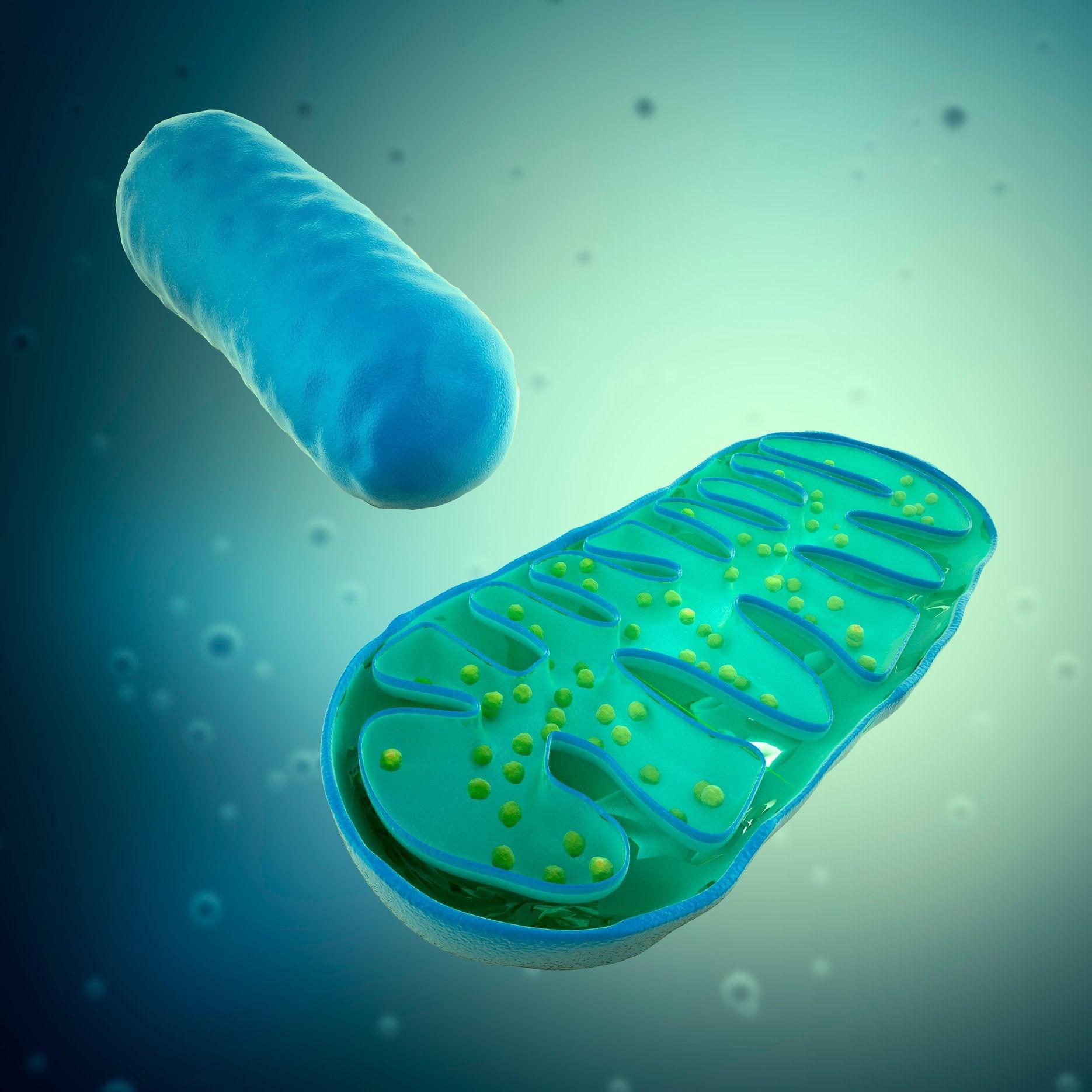
A Mitochondrial Mechanism Emerges
The most unexpected finding is where the mechanism resides. Transcriptomic and chromatin analyses reveal minimal differences between aged normal and MLKL-deficient stem cells.
This suggests that the primary driver is not gene regulation. Instead, the difference emerges at the level of organelles.
Mitochondria in aged stem cells become structurally abnormal and metabolically impaired. MLKL localizes to these mitochondria, where it reduces membrane potential and disrupts energy production. This provides a direct link between signaling and metabolic dysfunction.
Why This Changes the Model of Aging
Traditional models of aging emphasize passive accumulation of damage or gradual transcriptional drift. This study introduces a different framework.
Here, damage is actively generated by a stress-responsive pathway that remains engaged over time. MLKL acts as an intermediary, translating stress signals into mitochondrial dysfunction.
The consequence is a population of cells that survive but perform poorly. In stem cell systems, this distinction is critical, because dysfunction propagates downstream through entire cell lineages.
Where Uncertainty Remains
The findings are robust but not complete. The study is conducted in mouse systems, and translation to human hematopoietic stem cells remains to be validated.
The rescue effect is partial, indicating that additional pathways contribute to stem cell aging. MLKL is a contributor, not a sole driver.
Finally, the downstream cascade from mitochondrial damage to lineage bias is not fully resolved. The study establishes the connection but leaves intermediate steps open for further investigation.
What This Means for Longevity
This work expands the strategic landscape of longevity interventions. Instead of focusing solely on removing damaged cells or replacing them, it highlights the importance of preventing dysfunction at its source.
Targeting pathways like RIPK3–MLKL could reduce the formation of metabolically impaired stem cells. This approach differs from senolytics, which act after damage has accumulated.
It also reinforces the importance of mitochondrial integrity in aging. Maintaining energy production is not merely supportive but appears central to preserving stem cell function and immune competence.
Source
Yamada, Y., Yang, J., Saiki-Tsuchiya, A., Watanabe, Y., Koide, S., Murai, S., Sorimachi, Y., Fukuda, Y., Sumiyama, K., Sagara, H., Nakano, H., Takubo, K., Iwama, A., & Yamashita, M. (2026). Non-necroptotic MLKL function damages mitochondria and promotes hematopoietic stem cell aging. Nature Communications, 17, Article 2798. https://doi.org/10.1038/s41467-026-71060-4