Main Points
- A 2026 Nature Aging study links LINE-1 activation to cardiac aging via cGAS–STING signaling
- Evidence supports causal contribution in mice, not yet confirmed in humans
- Pharmacologic inhibition improved cardiac function, but specificity remains uncertain
- The work reframes aging as loss of genomic control triggering immune activation
Aging is often described as damage accumulating over time. But some of that damage may not accumulate at all. It may re-emerge.
Hidden within the genome are sequences that were never meant to be active. In youth, they are tightly suppressed. With age, that suppression weakens. The genome, in a sense, becomes less disciplined.
A recent Nature Aging paper suggests that in the heart, this loss of control is not just noise. It may actively push tissue toward dysfunction.
Why This Matters
Cardiac aging is typically approached through downstream risk factors like cholesterol, blood pressure, and fibrosis. These are measurable and actionable, but they don’t explain why aging itself predisposes the heart to failure.
This study proposes a different entry point. It suggests that retrotransposon activation, specifically LINE-1, may sit upstream of inflammation and senescence in aging cardiac tissue.
If correct, this reframes part of cardiovascular aging as a failure of genomic regulation, not just cumulative wear.
The Genome Is Not Static
A large fraction of the human genome consists of repetitive elements. Among them, LINE-1 retrotransposons retain the ability to copy and mobilize, at least in principle. In most cells, they are epigenetically silenced.
That silence is not permanent. Aging disrupts chromatin organization and DNA methylation patterns. As those controls weaken, LINE-1 can become transcriptionally active.
This is not merely a transcriptional curiosity. LINE-1 activity generates nucleic-acid intermediates that resemble viral material. The cell does not distinguish intent. It responds to structure.
When Self Looks Like Threat
Cells detect abnormal DNA through innate immune pathways, most notably cGAS–STING. This system evolved to identify infection, but it can also respond to endogenous DNA appearing in the wrong place.
When LINE-1 is activated, it can produce cytosolic DNA fragments. These fragments engage cGAS, which activates STING, triggering inflammatory signaling.
This creates a form of sterile inflammation, a known component of aging biology. In the heart, even moderate increases in inflammatory signaling can disrupt structure, function, and repair.
The key question is whether this pathway is incidental or causal.
Forcing the System to Break
To test causality, the researchers engineered mice lacking Mov10 specifically in cardiomyocytes. MOV10 normally helps suppress LINE-1 activity. Its removal creates a controlled loss of repression.
The resulting phenotype was not subtle. These mice developed early-onset cardiac dysfunction and aging-like features within months, far earlier than typical aging trajectories.
This does not prove that natural aging follows the same path. It does show that LINE-1 derepression is sufficient to induce key features of cardiac aging under experimental conditions.
Interrupting the Cascade
The study then tested whether the pathway could be interrupted. Two interventions were used.
The first, 3TC, inhibits reverse transcriptase activity and reduces LINE-1 propagation. The second, H-151, inhibits STING, targeting the downstream immune response.
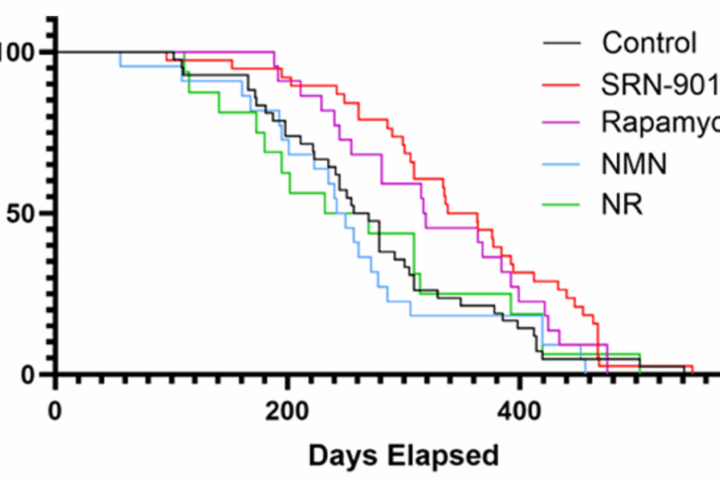
In aged mice, both interventions produced improvements in cardiac function. Measures such as LVEF and LVFS improved, alongside reductions in fibrosis, inflammatory markers, and senescence-associated proteins like p16 and p21.
The magnitude of improvement appears meaningful but not complete. These are partial rescues, not reversals.
What the Data Supports—and What It Doesn’t
The data support a mechanistic model in which LINE-1 activation contributes to cardiac aging through immune activation. The combination of genetic disruption and pharmacologic rescue strengthens that argument.
However, the pathway should not be interpreted as linear or exclusive. Aging involves multiple overlapping processes. LINE-1 activation likely interacts with other drivers, including mitochondrial dysfunction and proteostasis decline.
Additionally, pharmacologic specificity remains an open question. Both 3TC and H-151 have known targets, but off-target effects cannot be fully excluded in vivo.
Mechanism as Network, Not Chain
One of the more important contributions of this paper is conceptual. It links several hallmarks of aging into a connected system.
Loss of epigenetic control allows retrotransposon activation. That activation generates signals that engage innate immunity. The resulting inflammation contributes to senescence and tissue dysfunction.
This is best understood as a networked process, not a simple cascade. Feedback loops likely exist. Inflammation can further disrupt genomic regulation, amplifying the signal.
The implication is that aging is not just accumulation. It is destabilization of regulatory systems that normally suppress internal threats.
Where Uncertainty Remains
The study is limited to mouse models and in vitro systems. Human cardiac aging may involve similar mechanisms, but this has not been directly demonstrated here.
The Mov10 knockout model represents a strong perturbation. Natural aging likely involves more gradual and heterogeneous changes. The extent to which LINE-1 activation rises in human cardiomyocytes remains unclear.
There is also uncertainty around therapeutic targeting. Chronic suppression of retrotransposons or immune pathways may carry unintended consequences, particularly in host defense.
These uncertainties do not negate the findings, but they constrain how far they can be extended.
Longevity Implications
This work shifts attention upstream of senescence. Instead of focusing only on clearing damaged cells, it suggests that preventing the signals that generate those cells may be equally important.
It also introduces retrotransposon control and innate immune modulation as potential intervention points. These are not traditional longevity targets, but they are mechanistically grounded.
More broadly, the study reinforces a pattern emerging across geroscience. Aging processes often originate from systems that fail to maintain internal order. When regulation slips, the body begins reacting to itself.
The result is not chaos, exactly. It is misinterpretation.
Source
Yang, Y., Du, J., Liu, X., et al. (2026). Targeting age-related LINE-1 activation alleviates cardiac aging. Nature Aging, 6, 414–429. https://doi.org/10.1038/s43587-025-01056-0